The Complete Guide to Medical Device Carrying Cases: Protection, Compliance, and Best Practices | GADAO
Table of Contents
- Introduction: Why Medical Device Protection Matters
- Understanding Medical Device Case Requirements
- Compliance Standards for Medical Device Cases
- Design Principles for Medical Device Protection
- Material Selection for Medical Device Cases
- Real-World Case Study: Surgical Equipment Protection
- Comparison: Medical Device Cases vs. Generic Storage
- Key Success Factors for Medical Device Cases
- FAQ: Common Questions About Medical Device Cases
- Conclusion and Next Steps
Why Medical Device Protection Matters
Medical devices represent significant investments for healthcare providers, manufacturers, and professionals. Whether transporting surgical instruments, diagnostic equipment, or specialized medical devices, the protective case you choose directly impacts device integrity, patient safety, and regulatory compliance.
A compromised medical device due to improper storage or transport can result in equipment failure, inaccurate diagnostics, failed procedures, and ultimately, patient harm. Beyond safety concerns, regulatory bodies like the FDA and EU MDR require manufacturers to demonstrate that medical devices remain protected throughout their entire lifecycle, including storage and transport.
The Challenge of Medical Device Protection
Unlike consumer electronics, medical devices face unique protection challenges. Many medical devices contain sensitive components that require precise environmental control, including temperature and humidity management. Some devices contain sterile components that must remain uncontaminated. Others contain calibrated instruments that cannot tolerate shock or vibration.
A professional medical device case must address all these requirements simultaneously while remaining compliant with regulatory standards, maintaining sterility protocols, and supporting efficient workflows in clinical environments.
What You’ll Learn in This Guide
This comprehensive guide walks you through everything you need to know about medical device carrying cases. You’ll discover protection requirements specific to medical applications, compliance standards you must meet, design principles that ensure device safety, material selection criteria, and best practices from 20+ years of experience protecting medical equipment.
Understanding Medical Device Case Requirements
Unique Protection Challenges for Medical Devices
Medical devices present protection challenges that differ significantly from consumer electronics or industrial equipment. Understanding these unique requirements is essential for selecting or designing an appropriate medical device case.
Sterility and Contamination Control
Many medical devices must maintain sterile conditions or remain free from contamination. A medical device case must prevent dust, moisture, and microorganisms from contacting the device. This requires sealed compartments, antimicrobial materials, and cleanable surfaces that can be sterilized without damaging the case.
Environmental Sensitivity
Medical devices often contain sensitive components that require specific environmental conditions. A medical device carrying case must protect against temperature fluctuations, humidity variations, and environmental contaminants. Some devices require temperature-controlled storage; others require humidity control to prevent condensation and corrosion.
Calibration Integrity
Many medical devices contain calibrated instruments that must maintain precise accuracy. Shock, vibration, and impact during transport can affect calibration. A professional medical device case must provide shock absorption and vibration dampening to protect calibration integrity.
Regulatory Documentation
Medical device cases must support regulatory compliance. The case design, materials, and manufacturing processes must be documented and validated. A medical device case must be designed and manufactured according to quality management standards like ISO 13485.
Key Protection Requirements
A comprehensive medical device case must address multiple protection requirements simultaneously:
Requirement | Importance | Implementation |
Impact Protection | Critical | Shock-absorbing foam, protective bumpers |
Moisture Protection | Critical | Sealed compartments, moisture barriers |
Contamination Control | Critical | Sealed design, cleanable surfaces |
Environmental Control | High | Temperature/humidity management |
Sterility Maintenance | High | Antimicrobial materials, sterilization compatibility |
Calibration Protection | High | Vibration dampening, stable support |
Regulatory Compliance | Critical | ISO 13485, FDA, EU MDR documentation |
Ease of Use | High | Intuitive design, efficient workflows |
Concept – Visualizing Your EVA Case Solution
Once requirements are clearly defined, the project moves to the Concept phase, where design possibilities are explored and refined.
From Requirements to Visual Concepts
The Concept phase translates detailed requirements into visual design strategies. Your design team presents a formal Preliminary Design Review (PDR) featuring multiple distinct design approaches for your custom EVA case.
The Three-Concept Approach
Professional design teams typically present up to three distinct concepts, each exploring different aspects of your requirements:
Concept A: Lightweight and Portable
- Prioritizes slim profile and minimal weight
- Ideal for professionals who carry equipment frequently
- Uses lightweight EVA foam and compact compartmentalization
- Optimized for hand-carry and travel scenarios
- Best for: Field professionals, photographers, technicians
Concept B: Maximum Protection
- Focuses on maximum impact protection with rugged design
- Features reinforced bumpers and thicker foam padding
- Prioritizes durability over minimal weight
- Designed for harsh environments and intensive use
- Best for: Industrial equipment, sensitive instruments, high-value items
Concept C: Balanced Solution
- Combines portability and protection
- Introduces innovative opening mechanisms or material combinations
- Optimizes for both functionality and durability
- Often includes unique features like modular compartments or quick-access sections
- Best for: Professional operations requiring both protection and accessibility
What the PDR Includes
Each concept presentation for your custom EVA case typically includes:
- 3D Renders:Professional visualizations showing the case from multiple angles
- Internal Layout Options:Detailed compartment configurations and foam insert designs
- User Interaction Diagrams:How users will open, close, and carry the case
- Material Specifications:Recommended EVA foam types, densities, and exterior materials
- Cost Estimates:Preliminary pricing for each concept
- Timeline Implications:How each design approach affects the overall schedule
Selection and Refinement
Rarely does a client select one concept exactly as presented. Typically, feedback combines elements from multiple concepts: “We like the outer shell design of Concept A, with the internal organization of Concept C, and the quick-access feature from Concept B.”
Once a direction is selected, the design team consolidates these preferred features into a single, polished design. This refined design leads to the Critical Design Review (CDR), where all stakeholders approve the direction before moving to detailed engineering.
Timeline for Concept Phase
The refinement process from PDR to CDR typically takes 2-3 weeks after the preferred direction is selected. This timeline allows for:
- Design team refinement and optimization
- Client feedback integration
- Technical feasibility assessment
- Cost and timeline validation
- Preparation for detailed engineering phase
Compliance Standards for Medical Device Cases
FDA Requirements for Medical Device Cases
The FDA requires that medical device manufacturers establish and maintain procedures to ensure that medical devices remain protected throughout their lifecycle. This includes protection during storage and transport.
FDA Guidance on Medical Device Protection
The FDA’s guidance on medical device quality systems (21 CFR Part 11 and Part 820) requires manufacturers to:
- Establish procedures for protecting devices during storage and transport
- Document and validate the protective measures used
- Demonstrate that protective measures do not compromise device safety or effectiveness
- Maintain records of protective measures and validation
A professional medical device case must be designed, manufactured, and validated according to these requirements. The manufacturer must provide documentation demonstrating that the case protects the device without compromising its function.
EU MDR Compliance
The European Union Medical Device Regulation (EU MDR) imposes strict requirements on medical device manufacturers, including requirements for device protection during storage and transport.
EU MDR Requirements
EU MDR requires that medical device manufacturers:
- Establish quality management systems that include device protection procedures
- Conduct risk assessments identifying protection requirements
- Implement protective measures that address identified risks
- Validate that protective measures are effective
- Maintain documentation of all protective measures and validation
A medical device case designed for EU market distribution must be validated according to EU MDR requirements and must be manufactured according to ISO 13485 quality management standards.
ISO 13485 Quality Management
ISO 13485 is the international standard for quality management systems specific to medical device manufacturers. This standard requires that all components of a medical device, including protective cases, be designed, manufactured, and validated according to documented procedures.
ISO 13485 Requirements for Medical Device Cases
- Design and development procedures for the case
- Risk management and risk assessment
- Material selection and validation
- Manufacturing process documentation and validation
- Quality control procedures and acceptance criteria
- Sterilization procedures (if applicable)
- Traceability and documentation

Design Principles for Medical Device Protection
Compartmentalization and Organization
A well-designed medical device case provides organized compartments that keep each component secure and protected. Compartmentalization serves multiple purposes:
- Prevents component-to-component damage during transport
- Enables quick identification and access to specific components
- Supports efficient workflows in clinical environments
- Maintains organization during storage
Compartment Design Considerations
- Custom foam inserts that precisely fit each component
- Adjustable dividers for flexibility with different device configurations
- Quick-access compartments for frequently used items
- Secure retention systems that prevent movement during transport
Shock and Vibration Protection
Medical devices containing sensitive components require protection from shock and vibration. A professional medical device case must provide multiple layers of shock absorption:
- Outer protective shell that absorbs initial impact
- Shock-absorbing foam that dissipates energy
- Vibration dampening materials that reduce vibration transmission
- Stable support structures that prevent component movement
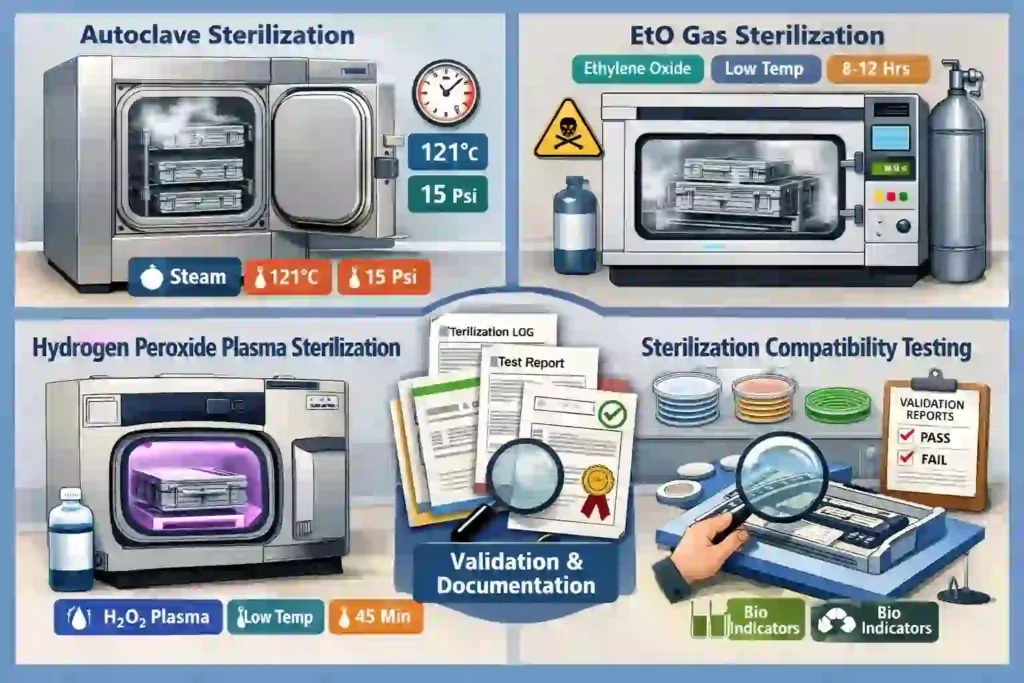
Cleanability and Sterilization
Many medical devices must be stored in sterile or clean conditions. A medical device case must support these requirements:
- Smooth, cleanable surfaces that resist contamination
- Sealed compartments that prevent dust and microorganism ingress
- Materials compatible with sterilization methods (autoclave, EtO gas, etc.)
- Antimicrobial materials that inhibit microbial growth
Environmental Control
Some medical devices require specific environmental conditions. A medical device case may need to:
- Maintain temperature within specified ranges
- Control humidity to prevent condensation or desiccation
- Protect against environmental contaminants
- Provide ventilation to prevent pressure buildup

Material Selection for Medical Device Cases
EVA Foam for Medical Device Protection
EVA (Ethylene-Vinyl Acetate) foam is the material of choice for medical device cases because it offers superior protection combined with regulatory compliance.
Why EVA Foam for Medical Device Cases
- Biocompatible:EVA foam is biocompatible and safe for contact with medical devices
- Customizable:EVA foam can be precisely cut and shaped for each component
- Sterilization Compatible:EVA foam withstands standard sterilization methods
- Antimicrobial Options:Antimicrobial EVA foam inhibits microbial growth
- Durable:EVA foam resists compression and maintains protective properties over time
- Cleanable:EVA foam surfaces can be cleaned and disinfected
EVA Foam Specifications for Medical Device Cases
Medical device cases typically use EVA foam with specific properties:
Property | Specification | Reason |
Density | 25-40 kg/m³ | Optimal shock absorption |
Hardness | 30-50 Shore A | Balance of protection and durability |
Thickness | 25-50mm | Adequate cushioning for sensitive devices |
Antimicrobial | Optional | Inhibits microbial growth in sterile environments |
Cleanability | High | Supports sterilization and cleaning |
Exterior Materials for Medical Device Cases
The exterior shell of a medical device case must provide protection while supporting regulatory compliance:
Hard Shell Materials
- Polycarbonate:Durable, transparent options available, good impact resistance
- ABS Plastic:Lightweight, durable, excellent chemical resistance
- Aluminum:Premium option, excellent durability, professional appearance
Soft Shell Materials
- Nylon:Lightweight, water-resistant, professional appearance
- Polyester:Durable, easy to clean, good chemical resistance
- Leather:Premium option, professional appearance, good durability
Hardware and Fasteners
All hardware and fasteners in a medical device case must meet regulatory requirements:
- Stainless steel fasteners for corrosion resistance
- Medical-grade zippers and latches
- Non-toxic adhesives and bonding materials
- Biocompatible materials throughout
Real-World Case Study: Surgical Equipment Protection
Project Overview: Hospital Surgical Equipment Cases
Client Profile: Major hospital system with 15 surgical suites requiring protection for specialized surgical instruments
Challenge: Protect expensive surgical instruments during transport between surgical suites while maintaining sterility and regulatory compliance
Solution: Custom EVA cases designed specifically for surgical instrument protection
Project Timeline and Results
Design Phase (4 weeks)
- Analyzed surgical instrument specifications and protection requirements
- Identified sterility and regulatory compliance needs
- Designed custom compartments for each instrument type
- Selected materials compatible with sterilization methods
Engineering Phase (8 weeks)
- Created detailed technical drawings and specifications
- Developed sterilization validation procedures
- Produced prototypes for clinical testing
- Refined design based on surgeon feedback
Production Phase (12 weeks)
- Manufactured 15 custom surgical instrument cases
- Validated sterilization procedures
- Completed ISO 13485 documentation
- Delivered cases with full compliance documentation
Total Project Duration: 24 weeks (6 months)
Measurable Results
Protection Performance
- Zero instrument damage during 18 months of intensive use
- Perfect sterility maintenance throughout storage and transport
- No contamination incidents
Operational Efficiency
- 30% reduction in instrument preparation time
- Improved surgical workflow efficiency
- Enhanced professional appearance in operating rooms
Regulatory Compliance
- Full FDA compliance documentation
- ISO 13485 certified manufacturing
- Sterilization validation completed
- Complete traceability and documentation
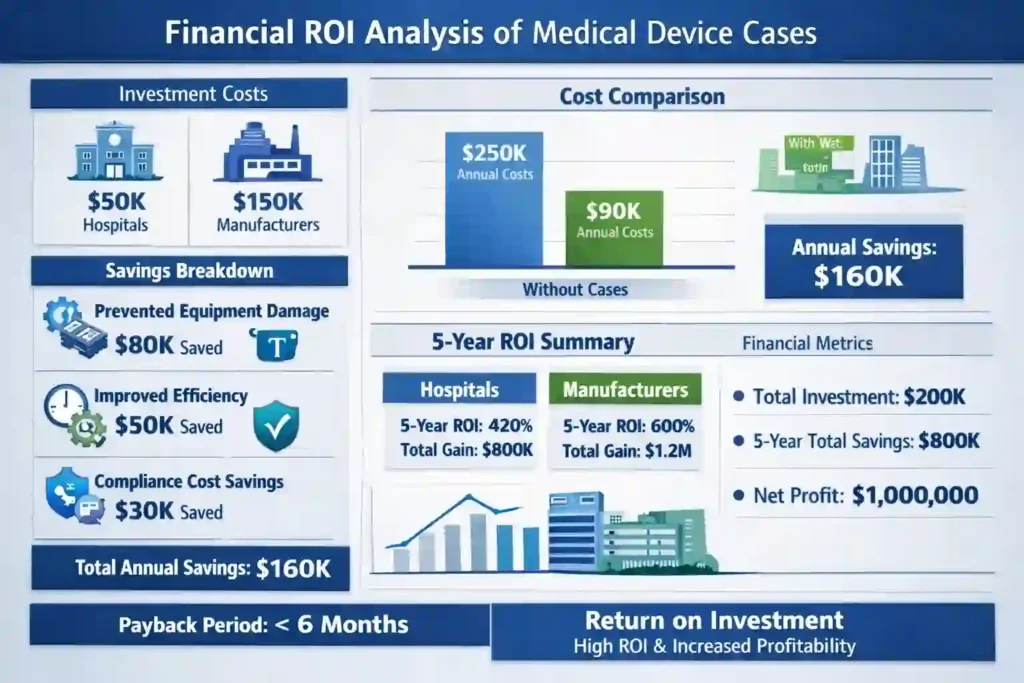
Financial Impact
- Initial investment: $45,000 (15 cases × $3,000)
- Prevented instrument damage (estimated): $150,000+
- Improved efficiency savings: $50,000+ annually
- ROI: 222% in first year
Comparison: Medical Device Cases vs. Generic Storage
Why Generic Cases Fail for Medical Devices
Many organizations attempt to use generic storage cases for medical devices. This approach creates significant risks:
Aspect | Generic Case | Medical Device Case | Impact |
Sterility Support | None | Full support | Contamination risk |
Regulatory Compliance | Not designed for | Fully compliant | Regulatory violations |
Material Safety | Unknown | Validated | Patient safety risk |
Sterilization Compatibility | Often incompatible | Fully compatible | Device damage |
Environmental Control | None | Optional | Device degradation |
Documentation | Minimal | Complete | Compliance gaps |
Cost | Lower initial | Higher initial | Lower total cost |
Total Cost of Ownership
While medical device cases have higher initial costs, the total cost of ownership is significantly lower:
Generic Case Approach
- Initial cost: $500 per case
- Replacement frequency: Every 2-3 years due to degradation
- Regulatory compliance costs: $10,000+ for documentation and validation
- Risk of device damage: $50,000+ potential losses
- Total 5-year cost: $15,000+ per case
Professional Medical Device Case
- Initial cost: $3,000 per case
- Replacement frequency: Every 10-15 years
- Regulatory compliance: Included in design and manufacturing
- Risk of device damage: Minimal
- Total 5-year cost: $3,000 per case
Key Success Factors for Medical Device Cases
- Early Compliance Planning
Start compliance planning early in the case design process. Identify all regulatory requirements before design begins to avoid costly redesigns.
Best Practice: Establish a compliance checklist covering FDA, EU MDR, ISO 13485, and industry-specific requirements before the design phase begins.
- Material Validation
All materials in a medical device case must be validated for biocompatibility and regulatory compliance. Never assume a material is safe without validation.
Best Practice: Request material safety data sheets (MSDS) and biocompatibility testing reports for all materials used in the case.
- Sterilization Compatibility
If the case will be sterilized, validate sterilization compatibility before production. Different sterilization methods (autoclave, EtO gas, hydrogen peroxide plasma) may affect different materials differently.
Best Practice: Conduct sterilization compatibility testing on prototype materials before committing to production.
- Cleanability and Maintenance
Design the case for easy cleaning and maintenance. Smooth surfaces, sealed compartments, and accessible areas support proper cleaning procedures.
Best Practice: Establish cleaning and maintenance procedures as part of the case design process.
- Documentation and Traceability
Maintain complete documentation of the case design, materials, manufacturing process, and validation. This documentation is essential for regulatory compliance and traceability.
Best Practice: Establish a document management system that tracks all design changes, material selections, and validation activities.
- User Training
Ensure that end users understand proper use, cleaning, and maintenance of the medical device case. Improper use can compromise device protection.
Best Practice: Provide comprehensive training and documentation to all end users on proper case use and maintenance.
FAQ: Common Questions About Medical Device Cases
Q: What compliance standards apply to my medical device case?
A: Compliance requirements depend on your specific application and target markets. Common standards include FDA requirements (21 CFR Part 820), EU MDR, ISO 13485, and industry-specific standards. Consult with your regulatory affairs team to identify all applicable standards.
Q: Can I use a generic protective case for my medical device?
A: While possible, using a generic case creates significant risks including regulatory non-compliance, material safety concerns, and potential device damage. Professional medical device cases are designed specifically to address these risks.
Q: How do I validate that my medical device case meets regulatory requirements?
A: Validation typically involves design reviews, material testing, sterilization compatibility testing (if applicable), and documentation of all protective measures. Work with your design partner and regulatory affairs team to establish a validation plan.
Q: What sterilization methods are compatible with EVA foam cases?
A: EVA foam is compatible with most sterilization methods including autoclave (steam), EtO gas, hydrogen peroxide plasma, and dry heat. Validate compatibility with your specific sterilization method.
Q: How often should I replace my medical device case?
A: Professional medical device cases typically last 10-15 years with proper maintenance. Replace cases if they show signs of degradation, damage, or loss of protective properties.
Q: Can I customize the appearance of my medical device case?
A: Yes! Medical device cases can be customized with logos, colors, and branding. Ensure that customization materials meet regulatory requirements for biocompatibility and safety.
Q: What documentation do I need for regulatory compliance?
A: Required documentation typically includes design specifications, material certifications, sterilization validation reports (if applicable), risk assessments, and quality control records. Work with your design partner to ensure complete documentation.
Q: How much does a custom medical device case cost?
A: Custom medical device cases typically cost $2,000-5,000+ per unit depending on complexity, customization, and regulatory requirements. Request a quote based on your specific requirements.
Conclusion and Next Steps
Medical device protection is not an afterthought—it’s a critical component of your quality management system and regulatory compliance strategy. A professional medical device case ensures that your devices remain protected, sterile, and compliant throughout their entire lifecycle.
Key Takeaways:
- Compliance is Essential:Medical device cases must meet FDA, EU MDR, and ISO 13485 requirements
- Material Selection Matters:Choose biocompatible, sterilization-compatible materials
- Design for Protection:Compartmentalization, shock absorption, and environmental control are essential
- Validation is Critical:Validate all protective measures before production
- Documentation is Required:Maintain complete documentation for regulatory compliance
- Professional Design Pays Off:The higher initial cost is offset by lower total cost of ownership
Ready to Design Your Medical Device Case?
GADAO brings 20+ years of experience designing medical device cases that meet FDA, EU MDR, and ISO 13485 requirements. Our expert team understands the unique challenges of medical device protection and specializes in creating compliant, effective solutions.
Next Steps:
- Schedule a compliance consultation
- Identify all applicable regulatory requirements
- Provide device specifications and protection requirements
- Receive a compliance-focused design proposal
- Begin the design and validation process





